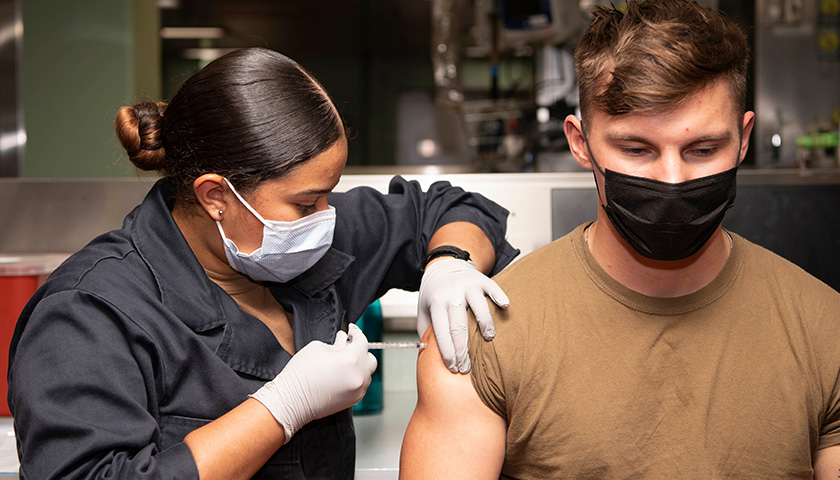
by Diana Girnita When the fear of getting COVID-19 was high and lock-down orders were in place, telehealth was an important resource, allowing patients to connect with doctors by live video, telephone, and remote patient monitoring without overcrowding hospitals and doctors’ offices. During this time of isolation and drastic increases in mental health challenges, telehealth services provided a lifeline of critical psychiatric and behavioral healthcare to people in need. Early in the pandemic, nearly half of all states and the federal government passed laws expanding access to telehealth. These changes allowed more providers to adopt this technology and, as a result, the United States went from having 43% of community health centers using telehealth before the pandemic to 98% just months into the pandemic. With innovation and quickly evolving technology, healthcare providers can deliver more high-quality services remotely, and our laws should make it easy to do so. Yet, as of last year, only eight states have made telehealth changes permanent. Congress passed the 2022 omnibus spending bill in March that extended federal telehealth provisions through September, the end of the 2022 fiscal year. The law covers telehealth visits, including video and audio-only visits, for Medicare patients. It also reinstated the CARES Act provision allowing high-deductible health plans (which…
Read the full story